
Bạn đang theo dõi
Lỗi khi theo dõi người dùng.
Người dùng này không cho phép người dùng khác theo dõi họ.
Bạn đã theo dõi người dùng này rồi.
Gói thành viên của bạn chị cho phép 0 lượt theo dõi. Hãy nâng cấp lên ở đây.
Bỏ theo dõi thành công
Lỗi khi bỏ theo dõi người dùng.
Bạn đã đề xuất thành công
Lỗi khi đề xuất người dùng.
Đã xảy ra lỗi. Hãy làm mới trang và thử lại.
Đã xác thực email thành công.


sanjay nagar,
india
Hiện ở đây đang là 12:36 CH
Đã tham gia vào tháng 9 16, 2021
0
Khuyến nghị
Vishnu K.
@Vishnu00542
0,0
0,0
0%
0%

sanjay nagar,
india
Không áp dụng
Công Việc Hoàn Thành
Không áp dụng
Đúng Ngân Sách
Không áp dụng
Đúng Thời Hạn
Không áp dụng
Tỉ Lệ Thuê Lại
Mechanical Design & Quality Assurance Engineer
Liên hệ Vishnu K. về công việc của bạn
Đăng nhập để thảo luận thông tin thông qua chat.
Portfolio
Portfolio


Sprocket Analysis
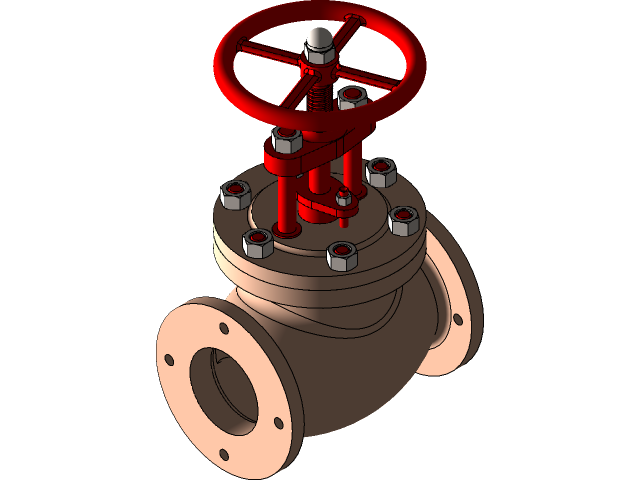

Stop Valve


Car Suspension


4 Cylinder Engine


Propeller
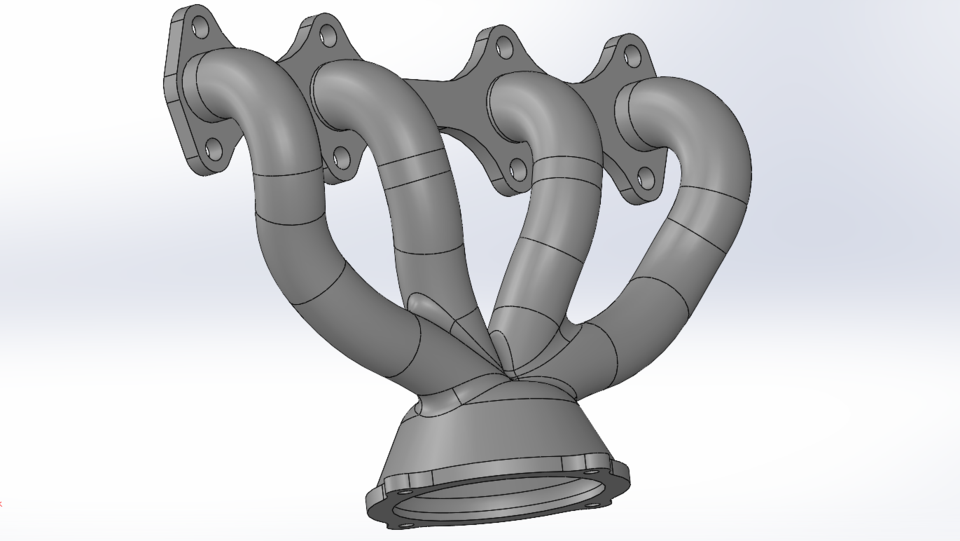

Intake Manifold


Sprocket Analysis


Stop Valve


Car Suspension


4 Cylinder Engine


Propeller


Intake Manifold
Nhận xét
Đã lưu thay đổi
Chưa có nhận xét nào để xem ở đây!
Kinh nghiệm
Mechanical Engineer
thg 5 2021 - Hiện tại
2 years of experience as Mechanical Engineer. Additional knowledge of EU MDR, IVD, US FDA, ISO 13485, ISO 14971, ISO 27001, etc,. for medical device industry products.
Working experience in Creo and SolidWorks.
Completed design and validation projects successfully.
Quality Assurance Engineer
2 years of experience as Mechanical Engineer. Additional knowledge of EU MDR, IVD, US FDA, ISO 13485, ISO 14971, ISO 27001, etc,. for medical device industry products.
Working experience in Creo and SolidWorks.
Completed design and validation projects successfully.
Học vấn
B. Tech
(4 năm)
Bằng cấp
The Project Management Course
Udemy
2021
1) Gained the project management knowledge and skills, necessary to manage an entire project
2) Review and understand a lot of project management templates and valuable documents.
3) Study Waterfall and Agile project management, performed with Scrum
4) Learned the fundamental theory and best practices of project management
Liên hệ Vishnu K. về công việc của bạn
Đăng nhập để thảo luận thông tin thông qua chat.
Xác thực
Các kỹ năng hàng đầu
Xem Các Freelancer Tương Tự
Xem Các Trường Hợp Tương Tự
Đã gửi lời mời thành công!
Cảm ơn bạn! Chúng tôi đã gửi email chứa đường link để bạn lấy tín dụng miễn phí.
Đã xảy ra lỗi trong khi gửi email của bạn. Hãy thử lại.
Đang tải xem trước
Đã cấp quyền truy cập vị trí.
Phiên đăng nhập của bạn đã hết hạn và bạn đã bị đăng xuất. Hãy đăng nhập lại.